Chapter 2: Lead white pigment in oil
FORMATTING POWERPOINT
#x INSTEAD OF 07 ETC
HART PROJECT 1: Oil & Pigment Preparation
Part 1: Background
Modern lead white versus traditional manufacture
Introduction
In 1893, after more than a century of experimentation had taken place with the most modern scientific methods for producing lead white pigment in Europe and the UK, a British Parliamentary Commission concluded that,
There is at present no substitute that can take the place of carbonate of
Lead made by the old Dutch process. (Lambert 1902 p.127)
As Thomas Lambert pointed out in Lead and Its Compounds (1902),
A white lead made by the old Dutch process is of the globular variety, and is found, when ground with linseed oil, to absorb more of the oil than the white lead, having a crystalline texture, and practice has shown that is has greater body and spreads better under the brush than the carbonates or sulphates of lead produced by precipitation in the former and by sublimation in the latter case. (p.126-27)
Lambert therefore concludes,
For this reason, all precipitated or sublimed lead pigments are considered inferior to genuine white lead. (p.127)
In fact, the search for a chemical process to manufacture lead white pigment more quickly, (and with lower toxicity to workers), has been one of science’s least successful endeavours, as chronicled by Homburg and Vlieger in their seminal article, “A victory of practice over science: The unsuccessful modernisation of the Dutch white lead industry.” (Homburg & Vlieger 1996).
Clearly, according to Lambert in 1902 and to Homburg & Vlieger in 1996, lead whites produced by sublimation or precipitation are significantly different and less desirable for paint making, than lead white resulting from the traditional stack (Dutch) process.
Scanning electron microscope backscattered electron images (SEM-BSE) of oil paint cross-sections prepared with modern lead white pigment compared with cross-sections from historical paintings (Powerpoint 01) immediately reveal that a commercially available modern lead white purchased in 1999 bears little physical resemblance to that used in the past. A comparison between a modern and traditional lead white pigments [1] in their dry state (Powerpoint 01) quickly reveals what Lambert was stating in the 1890s: their morphology is significantly different. Therefore, unless it was made by the traditional stack process (see below), lead white, the very basis of oil painting, had become significantly different, morphologically at least, by the late twentieth century.
Lead white pigment manufacture
Prior to the twentieth century the preferred method for producing lead white pigment, the stack process (or Dutch stack process), was to corrode lead metal slowly in the presence of dilute vinegar (acetic acid) using either horse dung or spent tanning bark as a source of heat and carbon dioxide (Powerpoint 01).
Differences in morphology in the modern versus traditional stack method are thought to be a function of the speed of corrosion and the specific corrosion products created. [2] Within the Dutch stack process method itself, crystal structure may also be influenced by changes in the process (e.g. the introduction of artificial heating), or in raw materials (Homburg and Vlieger 1996). In an early twentieth century source, it was noted that minor amounts of hydrogen sulphide from horse dung produced traces of lead sulphide (Petit 1907 p.15-16). Although replacing horse dung with spent tanners bark (the English method) eliminated this undesirable ingredient, one source reports that it meant that the
whole process took much longer: six weeks with horse dung, compared with 2-3 months with spent tan bark (Petit p.16).
Dutch stack process lead white is no longer made on an industrial scale.[3]Fortunately, a supply of lead white prepared in a reconstruction of the Dutch stack process was available for study and use by the HART Project [4] (see Suppliers). A comparison between this lead white, designated “Seynaeve” from the name of the supplier, and a sample of 18th century lead white [1] confirms that morphologically at least, the materials are very similar (Powerpoint 01).
Lead white pigment formulations
In HART Project 1.2 an investigation of various sources of lead white pigment was undertaken as part of the HART project and for research being carried out by Katrien Keune at FOM-AMOLF (Keune 2005 and Powerpoint 01.). Peter Hallebeek, senior conservation scientist at ICN, kindly carried out a study of pigments and oil paint using a Gadds Bruker/Siemens Microdiffractometer. Results are detailed in Chapter 2 Appendix I, and original graphs are in The HART Report Addenda.
Paint with modern versus Dutch stack process pigment
Morphology of stack process lead white
Cross-sections prepared by Kathrin Pilz (HART Project) of oil paint made with the reconstructed traditional stack process lead white (Seynaeve) consistently show a large range of apparent particle sizes: tiny particles (0.5μm) are intermixed with very large “particles” (20-40 μm). This is a feature of stack process lead white, as a similar range is seen in cross-sections of micro-samples from historical paintings (e.g. Rembrandt and Van Gogh, Powerpoint 01). Thanks to SEM-BSE imaging of ion polished oil paint cross-sections, Boon was able to demonstrate that the so-called range in particle size is due to pigment flocculation, as the larger “particles” actually consist of lumps of smaller
particles. [5] The grinding method has little effect on this tendency for stack method lead white to flocculate since both hand-ground and machine-ground reconstructions using Seynaeve lead white display this feature (see Chapter 4 Part 1, Section 2 Grinding Project and Powerpoint #21).
Handling qualities
Extensive experience with hand grinding two modern crystalline varieties of lead white pigment in linseed oil (Carlyle 2001b) formed the basis for comparing the behaviour of the reconstructed Seynaeve stack process pigment when ground with linseed oil extracted from the same seed lot used for the modern lead whites. The stack process pigment was markedly “softer” and less dense in feel under the granite muller than the modern equivalents. The stack process lead white formed a very responsive and delicate paint, which was highly thixotropic (it should be noted that modern hand-ground and machine-ground lead white in oil without extenders also display thixotropic behaviour).
A comparison of freshly milled Seynaeve lead white in poppy oil with a commercial artists’ lead white tube paint from Old Holland was striking: the commercially prepared paint felt harder, had more tack, and was less responsive to the palette knife than the Seynaeve lead white fresh from the laboratory roller mill. However, such a comparison must be tempered by numerous caveats: fresh paint versus paint prepared months before will likely be significantly different in feel, and the oil paint binder in the modern paint was not directly comparable to the pure poppy oil which was extracted directly from the seeds using the HART project oil press Chapter 2 Appendix III, and see below Part II Section A, Oil Washing. The terms adopted by the HART Project team to describe oil paint characteristics during grinding and painting-out are given in Chapter 4.
The presence of lead acetate
During manufacture, stack process lead white was normally washed in water during grinding to reduce the lead white flakes to powder. Water-washing also served to remove residual lead acetate formed during the corrosion process (Pulsifer 1888, Lambert 1902, Petit 1907). A British nineteenth century paint scientist, Sir Arthur Church, described lead acetate as an “accidental impurity” and provided a test for artists to establish whether it was present in their lead white in “objectionable proportion.”(Church 1890 pp.112-113). Thus, it was recognized that lead acetate could be present in the pigment due to insufficient washing.
Previous experience in the MOLART Fellowship project (Carlyle 2001b), with the deliberate addition of lead acetate (lead acetate trihydrate) to lead white pigment, as recommended by one 19th century British painter, indicated that this material has a profound effect on the texture, handling qualities and final colour of lead white paint. Powdered lead acetate was mixed with dry modern lead white pigment then both were ground together in linseed oil. The paint was very light, fluffy and “aerated.” It had a buttery short consistency, and when dry became significantly more yellow than comparable lead white paint without this addition (Carlyle et. al. 2002).
Lead Soap Aggregates
Background
The aggregation of lead soap in oil paint results in a significant paint defect found in many oil paintings from at least the 15th through to the 20th centuries on all types of support (Hermans 2017 pp.10-11). Thus far there has been no discernible relationship to the paintings’ age or country of origin. Aggregates which have protruded through the surface of the painting have a disturbing impact on the paint surface, causing changes in texture and appearance (Figs.1a&b). These aggregates, primarily carboxylates of palmitic and stearic acids are often associated with paint layers containing lead white pigment (Fig.1b) (see survey of literature on this in Hermans 2017 and recent work by Katrien Keune and Annelies van Loon). Judging from the relationship between the aggregates and the surrounding paints, it appears that the aggregates have formed in situ. The mechanism of their formation and what initiates aggregation of soaps (which are normally dispersed in the paint layers), was a topic of research within the De Mayerne Programme. [6]
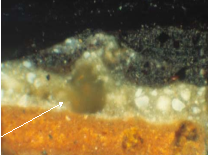
Images courtesy of the Conservation Department, Mauritshuis, Den Haag. Images and cross-section by Petria Noble.
Because lead soap aggregation does not follow any clear association with particular painting practices, nationality or era, at the time of the HART Project (2002-2005) it was felt by this researcher that a rather general aspect of the materials may be responsible, such as pigment or paint quality.
The quality of artists’ materials
Artists’ oil painting materials are generally processed prior to use (Carlyle 2001a & b). In many cases this involves washing pigments and oil in water to effect purification. Such pre-processing was time consuming and added to labour costs. It is not surprising therefore to find in 19th century British artist’s manuals and handbooks complaints of materials being insufficiently washed and therefore incompletely purified (Carlyle 2001a, Chapter 15, and insufficient washing of lead white, p. 513).
Since lead soap aggregates have been associated with paint layers containing lead white pigment, the role of purifying lead pigment with water-washing was examined. Similarly, prior to use, water-washing was used to purify linseed oil, a common binder in oil paint. Since lead soaps appeared to result from a combination lead white pigment and linseed oil, the effect of water-washing on the pigment as well as on the oil was explored. This project was broken down into two steps: water-washing linseed oil and water-washing Seynaeve stack process lead white pigment.
Part 2. Water-washing linseed oil, lead white pigment and oil paint preparation
The overall objective of HART Project 1 has been outlined above at the beginning of this chapter.
Section A: Oil washing procedure
Introduction
Traditionally, drying oils such as linseed oil were treated prior to use in oil painting to remove mucilage. This water-soluble component could be removed in a variety of ways; a traditional technique was to wash the oil with water. Mucilage left in the oil was reported to make it less clear, more coloured, and to slow the drying of the oil (Carlyle, 2001a p.31). Washing in water was described in early 17th century sources and throughout the centuries up to and including the 19th (see Stols-Witlox, Chapter 2 Appendix II Section 1). Other methods for removing the mucilage were used in the past. One was to allow the mucilaginous materials to settle out (precipitate) merely by standing. Alternatively, it could be removed by heating alone, or by treatment with litharge (lead (II) oxide) with and without heat. These latter methods were explored in the Carlyle MOLART Fellowship (Carlyle, 2001b [7]).
For the HART project a 19th century description of water-washing was chosen primarily because the same process was used in the Carlyle MOLART Fellowship (see recipe transcript Chapter 2 Appendix II Section 1). Some historical recipes for water-washing included salt, sand or breadcrumbs. Because the earliest research had found chlorides associated with protrusions in Rembrandt’s Dr. Tulp (Noble et al. 2000, Heeren et al. 1999, and Noble & Wadum, 1998), it was decided to evaluate the role of salt in water washing, to establish whether this could be a source of chlorides.
The Oil
For HART Project 1 the oil used for water-washing was extracted from the same seed lot: a supply of organically grown linseeds from the Electra cultivar (grown in the Netherlands). The seeds used had been acquired for the Carlyle MOLART Fellowship in 1999 (Carlyle 2001b). More information on these flax seeds can be found in Powerpoint 02: Visit to flax/linseed producer and in the visit description, Chapter 2 Appendix II Section 3. Details on the oil yield (% oil by weight of seeds) are in Chapter 2 Appendix III. After the 1999 supply of Electra seeds was exhausted, a second lot of Electra seeds was purchased from the same supplier in 2005 for other HART Projects (summarised in Suppliers).
For HART Project 1, oil from two extractions from the 1999 Electra seeds were used, one from December 1999 (referred to as “1999 oil”), and one which was freshly pressed in January 2003 (referred to as “2003 oil”), see Table 1. The method of oil extraction, and the equipment used in 1999 has been described in the Carlyle MOLART Fellowship Report (Carlyle 2001b). Illustrations are in Powerpoint 03: Oil extraction and water-washing. Because the oil yield was very limited with the small oil press used in the MOLART 1999 Fellowship, the HART Project had a custom-made oil press designed and built at FOM-AMOLF (all specifications are included in the oil press manual Chapter 2 Appendix III). The FOM-AMOLF stainless steel press was a highly effective and efficient means for extracting the oil.
An additional commercially produced oil (Table 1) was treated by water-washing for comparison. This unprocessed oil was obtained from a reconstruction of a traditional oil mill, De Bonte Hen in the Netherlands, and was designated De Bonte Hen B (see Suppliers and Powerpoint 03). De Bonte Hen B was pressed from Canadian seeds which were likely those bred for maximum oil yield. As it was felt that these seeds were not historically representative, they were not included in the lead white paint making experiment.
The preparation of the water
Tap water was passed through a deionising column, then through a “Millipore” water purifier, which produced “Type 1” purified water (Suppliers). Two salt solutions were prepared, one with laboratory grade sodium chloride (NaCl) and one with “sea-salt” (see Suppliers). Although prepared consistently each time, the salt-water solutions were not exactly 10% since the procedure was to measure 100 g. salt then to dissolve it in 1000 ml water (resulting in a total water-salt volume of 1026.2 ml NaCl, and 1025.00 ml of Sea-salt).
| Table 1: Electra seed oil and water used in the water-washing experiment | ||
|---|---|---|
| Oil Number | Oil extraction date | Water Used |
| 1 | 1999 December | Millipore |
| 2 | 2003 January | Millipore |
| 3 | 1999 December | Millipore + NaCl |
| 4 | 2003 January | Millipore + NaCl |
| 5 | 2003 January | Millipore + Sea-salt |
| 6 | De Bonte Hen B (2002 oil purchased) | Millipore + Sea-salt |
| 7 | De Bonte Hen B (2002 oil purchased) | Millipore |
| 8 [repeat of No.1] | 1999 December | Millipore |
| 9 [water not shaken or changed] | De Bonte Hen B (2002 oil purchased) | Millipore |
| 10 | De Bonte Hen B (2002 oil purchased) | not water washed |
| 11 | 1999 December | not water washed |
| 12 | 2003 January | not water washed |
The 1999 and 2003 extractions from the Electra linseeds were separated into a total of eight portions (highlighted in yellow Table 1). Both the 1999 and 2003 extractions were washed with Millipore water alone (#1 and #2) and with Millipore water plus sodium chloride (#3 and #4). The 2003 extraction and De Bonte Hen B were washed with Millipore water plus sea-salt (#5 and #6). De Bonte Hen B and another portion of 1999 oil and were washed with Millipore water alone (#7 & #8). One portion of De Bonte Hen B (#9) was mixed with water then left without further shaking and water changes to establish what effect these steps had on the purification process. Another portion of De Bonte Hen B was left untreated without water washing (#10) as were portions from both the 1999 and 2003 extractions (#11 and #12).
Glass separation vessels with a capacity of 250 ml (Powerpoint 03) were cleaned and dried prior to use. The ratio of oil to water was 1: 2 (50 ml oil to 100 ml water), based on the 19th century recipe (Chapter 2 Appendix II Section 1). Water in each vessel was replaced twice a week (on Mondays and Thursdays), and each vessel was shaken for 20 seconds each day (except Saturday and Sunday when the laboratory was closed).
To simplify the procedure and avoid mixing up the different waters and oils, in addition to their labels, each container of water was given a coloured dot with matching coloured dots placed on the corresponding separation vessels.
After 25 days, to encourage separation of the remaining mucilage the procedure was changed such that after water was removed twice weekly, the vessel was briefly inverted, then the remaining oil and mucilage were allowed to stand for 2.5 hours to effect complete separation. The mucilage which remained in the neck of the vessel was then removed. After this step fresh Millipore water was added and all ingredients were shaken together. Although no oil was visible in the mucilage at the time of removal, after standing, oil did separate out from the mucilage and was later recovered.
The washing procedure continued for a total of 7 weeks. All oils were then allowed to settle in the glass separation vessels. When no further separation of mucilage was visible, the oils were decanted into glass storage jars (see observations below). Thin polyester film (Mylar®, CCI stock, see Suppliers) was placed over the opening of each jar, and a plastic lid was screwed down. Jars were not completely full of oil therefore some air was present in each jar. Oils were stored in the laboratory in ambient light and temperature conditions.
Note: When oils were decanted, vessels #5 and #6 containing sea-salt treated water exhibited a significant build-up of salt crystals near the stopcock. Therefore, these oils were poured out through the top of the separation vessel to avoid any contamination from the salt residue.
After water-washing of the oils and leaving them to stand in their jars to ensure no further mucilage-water solution had separated, a drop of each oil was placed on a labelled glass slide (slides were previously cleaned with 100% ethanol) in order to observe their relative rates of drying (see Powerpoint 03).
Observations during water-washing
The amount of mucilage after 4, 18 and 25 days
After 4 days, a cloudy white material was visible in the neck of the separation vessel with oils being washed with sea-salt water. After 18 days this white cloudy solution was clearly evident in all the oils in their glass separation vessels, and by 25 days, the white solution was present in substantial amounts (Powerpoint 03). After 25 days, there was a clear line of separation from the oil, and the white solution was removed from each vessel. Observations on the state of the oils during washing are summarised in Chapter 2 Appendix II Section 2 Table 2.
The amount of mucilage after 25 days according to oil treatment
After 25 days of washing, the 2003 extracted oil washed with sea-salt treated water (#5) had a significantly greater amount of white material (mucilage) than the other linseed oils from Electra seeds. Next in quantity of mucilage was the oil extracted in 1999 washed with Millipore water alone. The 1999 oil with NaCl (#3) had very little mucilage present. However, it is interesting that of the De Bonte Hen B oils, it was the one treated with Millipore water only (#7) that had the greatest amount of mucilage, not the oil treated with sea-salt (#6) (Powerpoint 03).
Table 2: State of Mucilage in Oils after 25 days of washing
| Table 2: State of Mucilage in Oils after 25 days of washing | |
|---|---|
| Mucilage separated out and removed | Mucilage not clearly separated |
| #1 & #8 1999 Millipore | #2 2003 Millipore |
| #4 2003 Millipore + NaCl | #3 1999 Millipore + NaCl |
| #5 2003 Millipore + sea-salt | #7 De Bonte Hen B Millipore |
| #6 De Bonte Hen B Millipore + sea-salt | |
Observations after 35 days
After 5 weeks (35 days), although the De Bonte Hen B #7 had the most mucilage of all, there was still no clear separation between the mucilage-water solution and the oil. The #1 oil was very bright and clear, with #8 approaching it. The other oils were not particularly clear.
Observations in the 6th week
There was a marked development in the oils between observations on Thursday 13 February (35 days) and Monday 17th of February (39 days). Within these 4 days, all the oils had become significantly clearer, and in all cases the mucilage had separated. Even the #7 oil which had appeared as an “eggy” froth (having the appearance of whipped whole egg) for many weeks, showed a separation, with the mucilage taking up much less volume.
Final removal of mucilage and decanting the oils
At the end of the 7th week (27 February 2003) the water was poured off and the oils were left to settle as there was still some mucilage present with the oil. Although #8 oil exhibited enough mucilage to fill the neck of the separation vessel, by the next morning the mucilaginous material had reduced in volume, and water had separated out such that there were three distinct layers in the glass vessel: on top, clear oil, then a white cloudy material (mucilage), and at the bottom a watery turbid solution. By Monday, March 3rd the mucilage in the neck of #8 had reduced in volume again, and there was a larger volume of water.
By 6 March (56 days), all remaining water and mucilage were removed from the oils, and on 18 March (68 days), the oils were decanted into fresh jars.
Throughout the experiment, wash water from each separation funnel was first poured into a glass beaker prior to being discarded. Over the weeks, a substantial clear sticky film was deposited on the glass. Some of the film was scraped off the glass and placed in a small glass vial should it be required for analysis in the future.
Results
The effect of not shaking or changing the water
In order to evaluate the impact of regular water changes and daily shaking of the oil and water, the same proportion of Millipore water as used throughout was added to De Bonte Hen oil B (#9) and the oil and water were left undisturbed. Mucilage separated out and within 4 days appeared very frothy. After 46 days the oil and mucilage were clearly separate (Powerpoint 03) and both were poured into a separation vessel. Ten days later water was removed leaving a large amount of curdled mucilage in the neck of the vessel. After 68 days the oil was still depositing mucilage and the oil itself was significantly less clear and more coloured than the same oil which had been treated by shaking daily with twice weekly water changes. Changing the water and decanting accumulated mucilage regularly resulted in oils with a superior appearance and with virtually all mucilage removed.
The age of the oil
Authors in the documentary sources studied valued aged linseed oil, reporting that the oil improves by age and by “repose” as simply allowing the oil to stand undisturbed was held to result in separation of the oil from its mucilage (Carlyle 2001a p.32). Oil which had been extracted in 1999 (#1) was compared with oil from the same seed lot freshly extracted in 2003 (#2). After 25 days more mucilage was visible in the 1999 oil than in the 2003 oil (Chapter 2 Appendix II Section 2 Table 2). At that time, as it was not possible to clearly distinguish between mucilage and oil in the 2003 oil (#2) therefore the mucilage could not be removed. After 35 days the 1999 oil (#1) was markedly clearer and more brilliant than the 2003 oil (#2). After 68 days, both oils appeared equally clear and were a similar light lemon colour. What is suggested by the behaviour of the 1999 oil is that the older oil loses its mucilage more quickly than the freshly extracted oil. This implies that older oil would have been ready for use earlier than the freshly pressed oil. This observation requires further research since the oils in this project were not specifically studied with regard to the amount of mucilage produced and the length of time to achieve clearance.
The addition of salt to the wash-water
Old oil (1999 extraction)
Adding pure sodium chloride (NaCl) to the oil extracted in 1999 (#3) did not improve the clarity or colour. In fact, at the completion of washing (68 days) the oil washed with NaCl treated water remained slightly turbid. During washing, there was no clear separation between the oil and the mucilage after 25 days so it was not possible to pour off the mucilage at this stage. This implies that the addition of sodium chloride to the oil did not hasten the removal of the mucilage (compare with the same oil washed in Millipore water alone, #1 above).
Freshly pressed oil (2003 extraction)
Adding salt as pure sodium chloride (NaCl) or as sea-salt did appear to aid the separation of mucilage from freshly extracted oil. After 18 days, while the 2003 oil with Millipore water alone (#2) appeared turbid, that with NaCl (#4) and with sea-salt (#5) showed clear separation of oil and mucilage with the #4 oil being very clear. After 25 days the freshly extracted oil with Millipore water only (#2) was still not separate from its mucilage (Chapter 2 Appendix II Section 2 Table 2). The oil with sea-salt treated water (#5) had a very large proportion of mucilage present, implying that the sea-salt was more efficient in removing the mucilage. However, although salted water appears to have hastened mucilage separation, interestingly, the oil washed in Millipore water only (#2) appeared brighter and clearer after the completion of washing than the oils washed with salted water.
The source of linseeds
There was a distinct difference in the colour and mucilage content in the oils derived from separate seed lots. De Bonte Hen B washed with Millipore water (#7) produced significantly more mucilage than the 1999 and 2003 extractions from the MOLART Electra seed lot which were washed in Millipore water (#1 & #2). Adding sea-salt to De Bonte Hen B helped to separate the mucilage more quickly (compare #6 & #7 after 25 days, Chapter 2 Appendix II Section 2 Table 2). Ultimately, after 68 days De Bonte Hen B washed with sea-salt treated water (#6) was similar in clarity to the freshly extracted MOLART oil washed with salted water (2003, #4 & #5). This oil (#6) was much lighter in colour than the De Bonte Hen oil washed with Millipore water alone. However none of the De Bonte Hen samples (#4, #5, & #6) were as clear as the 1999 oil (#1 & #8), and the colour of De Bonte Hen oil remained tawny to orange-yellow, quite distinctly more coloured than the pale lemon yellow of the oil from the Electra seeds (Powerpoint 03).
Mucilage separation
Although some oils appeared brilliant and clear after only 5 weeks (especially #1 the 1999 oil washed in Millipore water only), white material was still separating from the oil as late as 7 weeks, indicating that mucilage clearance was not completed despite the appearance of the oil. The sixth week of washing was significant, as this was the point of separation for any white material which had not separated from the oil previously. It appears that in the latter stages of washing the mucilage or white material in the oil was present in a lower proportion in the water. This was evident when, in the last stages of water-washing, the white material separated out into two phases — a large amount of water and a very small amount of white material (Powerpoint 03).
At the beginning of the washing procedure, the mucilage removed from each oil was discarded. Later on it was saved and was found to separate into oil, water, and a yellowy-white material. Since the oil recovered from the mucilage was very clear, it was added back to the original portion in the separation vessel. In future, mucilage from all stages of washing should be kept to determine whether oil can be recovered.
Generally, for each oil, between 7 to 10 ml of oil was lost during washing. This is an estimate only as oils were not weighed before and after washing, as they should be in any future experiments of this type.
Drying time of water washed oils
After being dropped into the centre of clean glass slides, De Bonte Hen B, washed with Millipore water (#9) and untreated (#10), spread out and seeped into the paper labels adhered across the bottom of each slide. This suggests that both of these oils had very low surface tension. The other oils from the Electra seeds extracted using the HART oil press all formed small puddles which eventually dried with a heavily wrinkled surface (Powerpoint 03).
The oils were applied to glass slides on 18 March 2003. By May 1 (approximately 6 weeks) the untreated 1999 oil (#11) and 2003 oil (#12) were still completely liquid whereas the other oils (#s 1 to 8) had developed wrinkled surfaces. Nineteen days later (20 May) the untreated 1999 oil (#11) had developed wrinkles over almost all of its surface, while the untreated 2003 oil still appeared completely liquid (Powerpoint 03). Eventually the untreated oil extracted in 2003 dried to form a wrinkled surface similar in appearance to the other oils.
Discussion
Washing oils with water does result in a less coloured, brighter and more clear oil. Steps such as daily shaking of the oil and water and twice weekly water changes with removal of accumulated mucilage contribute to this, as oil left on water without these steps did not result in complete elimination of mucilage and a completely clear oil. The mucilage in oil which was freshly expressed does not separate out as quickly as mucilage in oil which had been extracted some 3.5 years previously. Linseed oils from the two different seed lots (Electra and De Bonte Hen B) do exhibit significant differences in colour, clarity and mucilage content.
Washing with water treated with salt gave inconsistent results. Freshly pressed oils (2003 and De Bonte Hen B) did appear to separate from their mucilage more quickly when salt was added to the water, however the end product was slightly less clear than when the oil was washed with Millipore water alone. The old oil (1999) did not benefit visibly from the addition of salt to the washing water since mucilage did not separate more quickly, and like the other salt-water treated oils, the end product was less clear than when the oil was washed with Millipore water alone. The use of sea-salt did result in a slightly greater quantity of mucilage in the separation vessels, but the end results did not appear more clear or less coloured than when water with pure sodium chloride was used, or when Millipore water was used.
Conclusions
It is unclear from this experiment whether the method of oil extraction plays a role in the amount of mucilage produced as the extraction methods were completely different for the two oils. The De Bonte Hen B oil was extracted in a full-size wooden reproduction of an early oil mill, whereas the HART Project oil was extracted with a laboratory-sized stainless-steel press (Powerpoint 03). It is interesting to note that a previous study found that preheating the seeds followed by cold pressing resulted in higher mucilage content than preheating followed by warm pressing (Riep 1936).
Section B: Pigment washing procedure
For pigment washing, flakes of lead white freshly prepared by Jef Seynaeve using the reconstructed stack method (Powerpoint 01) were used for the pigment washing procedure. Flakes of white lead acquired in October 2002 and designated “Seynaeve Batch 1” were ground to a uniform powder with a ceramic mortar and pestle. A 1000 ml glass beaker was filled with 250 g of this lead white powder, then Millipore water was added up to the 800 ml mark on the beaker (Powerpoint 04: Pigment washing, grinding & substrates). The lead white and water were stirred with a glass rod for approximately one minute then allowed to settle. A watch-glass was placed over the top of the beaker for dust protection, and the beaker left undisturbed.
Water-washing schedule
Beginning the next day and continuing for a total of ten days: water was removed from the beaker daily using a 100 ml glass pipette introduced with the tip situated near the centre of the water (500 ml mark on the beaker). Fresh Millipore water was added up to the 800 ml mark, the lead white and water were stirred vigorously with a glass pipette for 60 seconds (using a timer), and then left to settle.
pH measurements
During water extraction, water was placed into three jars. The 3 examples of wash-water were then taken to a pH meter and measurements were taken immediately. The pH meter was calibrated before each set of readings with buffer solutions of pH 7 and pH 4 (Millipore water alone was pH 6.7). Initially 3 measurements from each wash-water were taken, then an average was taken from all 9 readings. Eventually, one reading from each wash-water was taken with the average taken from those 3 readings (Table 3).
Completion of washing
After 10 days of daily water changes, the lead white pigment and Millipore water were left undisturbed for a further 5 days; then pH readings were taken and the wash-water changed. A week later another set of pH readings were taken, the water was changed again, and then 3 days later the pH of the wash water was measured. It appeared that no further change was taking place, therefore the final wash-water was drawn off, and the wet pigment placed in a wide-mouthed glass jar to dry. The jar was covered with a sheet of polyester film held above the opening with a glass rod placed over the mouth of the jar to allow air circulation while discouraging dust.
Table 3: Average of pH readings of lead white wash-water
| Date | pH | Date | pH |
|---|---|---|---|
| Day 1. Tues. Jan 28 | 5.89 | Day 8. Tues Feb 4 | 6.17 |
| Day 2. Wed Jan 29 | 5.83 | Day 9. Wed Feb 5 | 6.22 |
| Day 3. Thurs Jan 30 | 5.86 | Day 10. Thurs Feb 6 | 6.28 |
| Day 4. Fri Jan 31 | 5.93 | Average of 3 readings | |
| Day 5. Sat Feb 1 | 5.98 | Tues Feb 11 | 6.25 |
| Day 6. Sun Feb 2 | 6.04 | Tues Feb 18 | 6.18 |
| Day 7. Mon Feb 3 | 6.09 | Fri Feb 21 | 6.23 |
Each time the wash-water was changed, a small amount of lead white was taken out of the jar and placed on a piece of polyester film with the date marked on the film. Once dried these samples were placed in glass jars.
Katrien Keune (FOM-AMOLF) investigated unwashed and washed samples of Seynaeve lead white and determined that lead acetate was being removed during washing. This was further confirmed during an investigation of these samples using solid-state Nuclear Magnetic Resonance. The effect of water-washing Seynaeve lead white pigment in terms of its composition and particle size was investigated in Stols-Witlox et al. 2012.
Section C: Lead white oil paint preparation
Procedure
A total of 28 oil paints were prepared using water-washed oils and untreated oil (see summary Table 1). The paints were divided into sets according to the oil used (Table 5a). The oil treatments selected were as follows: washed with Millipore water alone (#1 & #2); Millipore water + pure sodium chloride (#3 & #4); Millipore water + sea-salt (#5); and untreated oils, one extracted in 1999 (#11) and one extracted in 2003 (#12). The oils are summarised in Table 4, and the paints in Table 5a.
Table 4: Oil pre-treatment by water-washing used for lead white paint
| Oils | Millipore Water | Water + NaCl | Water + Sea-salt | Untreated |
|---|---|---|---|---|
| 1999 Oil | #1 | #3 | #11 | |
| 2003 Oil | #2 | #4 | #5 | #12 |
The lead white pigment came from Seynaeve Batch 1 (see Suppliers). One portion was washed in Millipore water (as detailed in Part 2 Section B above), and one portion was left unwashed. Of the two portions of lead white (washed and unwashed), two sets of paints were made — one dry-ground, and the other wet-ground. That is, one of the paints was ground with dry pigment and oil; and the other with pigment which had been ground first with Millipore water, prior to the addition of oil. This latter step was included because some early instructions for grinding lead white called for the pigment to be wet first with water (Chapter 2 Appendix I).
Table 5a details the amount of pigment and oil used in each paint. Each of the 28 paints was prepared by hand-grinding on a granite slab with a granite muller (see description Chapter 2 Appendix IV).
Oil paint preparation
Oil paints were prepared between March 24th and April 3rd 2003, taking a total of 8 days for all 28 paints to be hand-ground and applied to the substrates (see Chapter 1 Appendix Master List of HART Projects). Each paint was applied to its set of substrates (Table 5b) immediately after it was ground (Powerpoint 04). Paint within each set was prepared in a standard order to minimize contamination from residue on the granite slab from previous grinding. Therefore, the washed pigment was ground first, followed by the unwashed pigment:
- Washed pigment, dry
- Washed pigment, wet
- Unwashed pigment, dry
- Unwashed pigment, wet
The order of preparation is not reflected in the unique numbers assigned to each paint (see summary Table 5a).
Generally, a single set, consisting of 4 paints, was made in a day. Observations on the handling characteristics of each paint were recorded during grinding and application to the substrates (Chapter 2 Appendix IV Tables 1–7, Summary Tables 8 & 9).
The amount of oil used
A fixed amount of pigment (12.5 g) was used throughout. Each paint was prepared by grinding pigment with oil such that the paint was as stiff as possible in order to establish an end point for grinding. Once the amount of oil needed for a given set was established, an attempt was made to use a consistent amount of oil for that set. However, it should be noted that the method of measuring the oil for this project using a glass 10 ml graduated cylinder was inefficient for small amounts of oil, so that oil measures can only be approximate to ±0.2 to ±0.3 ml (later on, HART projects relied on weighing the oil to provide greater accuracy).
Table 5a: HART Project 1 — Lead white washing and oil washing experiment
(Listed in order of preparation)
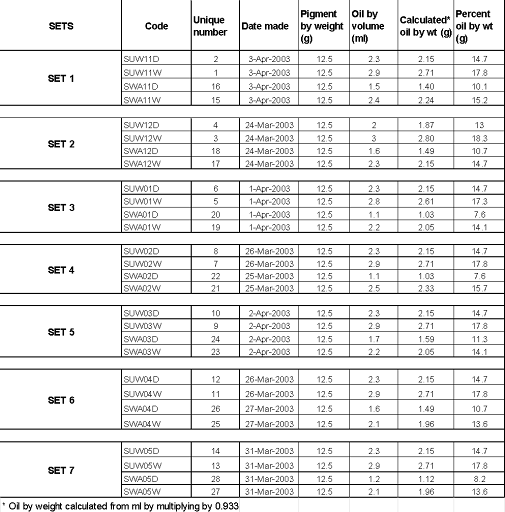
For detailed information on substrates and oils, see Chapter 1 Appendix Master List of HART Projects, HART Project 1.
Hand-grinding pigment, dry and wet
The procedure for hand-grinding lead white paint with dry pigment is described in Chapter 2 Appendix IV Section 1.
The procedure for hand-grinding with wet pigment was based on references to this method in historical documentary sources (Chapter 2 Appendix I). The dried pigment (12.5 g) was first crushed on the slab with the muller, then ground dry under the muller to achieve a fine even powder. The powder was gathered to the centre of the slab using a stainless steel scraper or the edge of a stainless steel palette knife. Millipore water (5 ml) was added at once to the dry powder and mixed slightly with the palette knife. The pigment and water mixture were then ground together with the muller in the same manner as grinding in oil. Very quickly a uniform creamy slurry resulted which was very light and quite stiff (rather like heavy cream in the final stages of whipping), see Powerpoint 04.
Once the amount of oil required for wet grinding had been established (by adding oil gradually to the wet slurry with continuous grinding) it was possible, with later paint samples, to add a high proportion of oil at once, reserving only a few ml to be added at the latest stage of grinding. During wet grinding, the paint goes through stages (detailed below under Observations, wet versus dry grinding). The procedure used for hand-grinding HART oil paint is detailed in Chapter 2 Appendix IV, see also Chapter 4 Terminology Paint Handling Characteristics.
The substrates
The paints were applied to a total of 9 substrates (Table 5b): 3 drawdowns onto polyester film (Melinex 0.10 cm thick, 10 × 16 cm width by length); plus 1 polyester film with excess paint (brushed/palette knife); 2 paint-outs on artists’ board; and 2 drawdowns on glazed ceramic tiles (one white, one black). As well, one small paint brushstroke was applied onto canvas with a flour-paste ground layer previously applied. The substrates are illustrated in Powerpoint 04 and detailed in Suppliers as well as in Chapter 1 Appendix I: Master List of HART Projects. The canvas strips with flour-paste are detailed in Chapter 3 Appendix VI and Powerpoint 19.
Each substrate was assigned a number which appears at the end of the paint code. For example, in paint code SUW11W-1.1, the last number (.1) indicates the substrate:
Table 5b: Substrates for HART Project 1 paint-outs — lead white & oil washing

Non-porous substrates were cleaned prior to use in the following manner: polyester sheets were wiped down thoroughly with ethyl alcohol using Kimberly-Clark Kleenex brand tissue (Supplier); ceramic tiles were washed with water and Fisher brand “Sparkleen” detergent (Supplier), then rinsed in tap water followed by Millipore water. Ceramic tiles received a final cleaning with ethyl alcohol using the same brand of tissue used for the polyester sheets.
Paint application
Brush and palette knife
Paint was applied to the commercially prepared artist’s boards and to one of the polyester films using an artist’s palette knife and an artist’s bristle brush (one brush was used for each paint set and was wiped clean between paint applications within that set). Paint was applied to the canvas substrates with an artist’s bristle brush (Suppliers).
Drawdowns
At the outset of the HART Project, the amount of Seynaeve Batch 1 pigment was limited (approximately 1 kg). As a consequence, drawdowns with a narrower width than those provided with a commercial drawdown bar were needed. Therefore, a special narrow-width (2 cm) stainless steel drawdown bar with a gap of 0.025 cm was custom-made at FOM-AMOLF for this project (illustrated in Powerpoint 04). Commercially available drawdown bars are approximately three times wider (6 cm) and therefore require more paint for a single application.
Section D: Light exposure and oven exposure
For each paint set, drawdowns on polyester film (designated as .1 and .2 substrates) were selected for an artificial aging experiment. Drawdowns on substrate .1 would be first exposed to light at the SRAL facility in Maastricht, followed by oven exposure at ICN (Suppliers). Drawdowns of the same paint sets on substrate .2 were controls, placed in copper-treated plastic bags to reduce oxygen exposure during the time that .1 drawdowns were being artificially aged.
Light exposure
Drawdowns .1 were sent to the SRAL facility. For transport, each sample was interleaved with cleaned polyester film to protect the paint surfaces and reduce chances of cross-contamination between the samples.
The paint samples were exposed 24 hours a day in the SRAL light-aging facility from 20 June through to 8 September 2003 (11 weeks). Samples were rotated so that they each received 8000 lux (daylight fluorescent tubes, Philips 96.5). The temperature ranged between 20–25°C, and the relative humidity (RH) was between 40–45%.
While the light-exposed drawdowns were being cut into strips in preparation for oven exposure (see below), a marked difference was observed in their response to being cut, with some samples exhibiting brittle behaviour (HART Project Laboratory Notebook Vol I p.99, Sept 9, 2003), summarised in Table 6.
Table 6: Descriptions of light-exposed paints during cutting

*Note: It was unclear whether samples 1, 2, and 3 were brittle, since they were not individually assessed — only that sample 4.1 was much more brittle than samples 1–3. It is assumed that samples 1–3 were somewhat brittle. Samples without a descriptor (blank) are assumed to have been not brittle.
Although incomplete, these observations on the relative brittleness of the samples from HART Project 1 exposed to light for 11 weeks indicate trends:
- Grinding type: Grinding the pigment in oil while the pigment powder was dry (dry grinding D) appears to result in paint that is more brittle than the same paint prepared with the pigment ground first in water prior to the introduction of oil (wet grinding W).
- Pigment treatment: Another trend appears to be related to whether or not the lead white pigment was water-washed prior to use (UW = unwashed, versus WA = washed). Generally, samples of paint with unwashed pigment (UW) were observed to be more brittle than those where the pigment was water-washed prior to use (WA). The exception is sample 20, with washed lead white, which was reported to be the “most brittle yet.”
- Oil processing: In terms of the oil processing method used (Sets 1 to 7 each use Electra linseed oil that was extracted at different times and either left untreated or water-washed with and without two types of salt), as evident in Table 6, there does not appear to be a correlation between the presence or absence of brittle behaviour during cutting according to the oil processing method.
As will be seen, after oven exposure the samples exhibited a range of colour changes from mild to severe, not only according to the length of oven exposure but also in relation to the sets made. Since the location of the samples during light exposure was not tracked, whether some samples received more or less light is not known, and the conditions in the oven may not have been completely uniform. Another factor to consider is that in these first two HART Projects, oil was measured by volume during paint-making, a significantly less accurate method than by weight (subsequent projects all measured oil by weight).
It is interesting to note that some of the samples which underwent oven exposure (below) were exhibiting extreme brittleness in 2020 (Powerpoints 06 & 07).
Oven exposure
The procedure was to expose paint samples for varying amounts of time in an oven at ICN at 50°C and 80% relative humidity (RH).
As noted above, to facilitate removal at regular intervals, prior to placing the light-exposed samples into the oven, each drawdown in set .1 was cut into 7 strips. One piece was held back as a control (with the original printed label as identification), and the remaining six strips of paint on their original Melinex® substrate were labelled with code extensions i–vi (Table 7).
Table 7: Oven exposure time
Oven Sample extension no. | i | ii | iii | iv | v | vi |
Exposure time (in days) | 3 | 6 | 10* | 20 | 40 | 70 |
*sample set iii was removed from the oven after 11 days, not 10 days as planned.
[1] A sample of historical lead white pigment dated to 1704 was kindly supplied to the project by Lisa Wagner, from the Vigani Collection, Queens’ College, University of Cambridge, UK. See Wagner (2007).
[2] Regarding the scale of the operation, Homburg states: “…in the original stacks there always were a few thousand of pots, in at least 5-7 layers. As a result of this configuration the fermentation of the dung was such that the temperature gradually reached a level of about 60-70 degrees centigrade for several weeks. The lead sheets corroded completely in this way. The fermentation lasted many weeks. As the size of the white lead crystals depends on the length of the reaction time and on the temperature, and therefore on the size and shape of the stacks, one can never be completely sure whether the experiments on a small scale produce white lead which is completely identical with the former industrial product (old Dutch method).” Correspondence with Professor Dr Ernst Homburg, Department of History, Faculty of Arts and Culture, University of Maastricht with Carlyle (16.10.2002).
[3] Specialist suppliers have since been making stack method lead white for artists: Natural Pigments (www.naturalpigments.com) and Michael Harding (www.michaelharding.co.uk).
[4] Paintings conservator and painter in Belgium, Jef Seynaeve, made small amounts for his own use (using horse dung) and was kind enough to supply the HART Project. According to Petit, dung from carnivores (e.g. pigs) was not used due to its significantly higher yield of hydrogen sulphide in the pigment [lead sulphide is a brown to black colour] (Petit 1907 15-16).
[5] Pigment flocculation was identified by Boon using ion polishing on micro-samples of lead white oil paint (Carlyle et al. 2008a). Flocculation points to a lack of complete dispersion of the lead white pigment in oil. The Kremer Pigmente lead white (#46000 purchased 1999) used in the Carlyle MOLART Fellowship and HART Project was likely treated with dispersal agents since oil paint samples viewed with SEM-BSE imaging do not show similar evidence of flocculated pigments (Powerpoint 01). For modern pigment surface treatments see Salis Gomes et al.(2020).
[6] Specifically: Imaging Microscopy of Paint Cross-sections (De Mayerne Project P4), The coordination chemistry of lead and chemical changes in ageing oil paintings (De Mayerne ProjecP3), The Painting Technique of Vincent van Gogh (De Mayerne Project P6), as well as the HART Project (P5).
